Increase Customer Satisfaction
Deliver zero-defect products, achieve optimal levels of customer satisfaction with QMS software.
Reduce Cost of Quality
Automate quality processes and instantly access to actionable data.
Simplify Audits
Our quality control software for manufacturing is a single source of truth to simplify regulatory & customer compliance reporting.
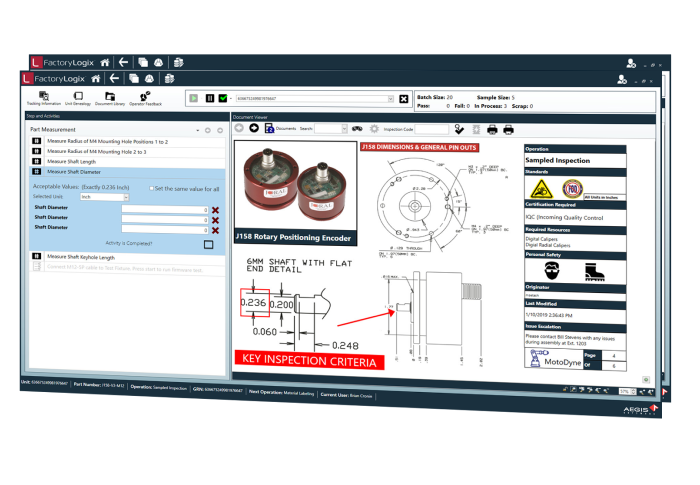
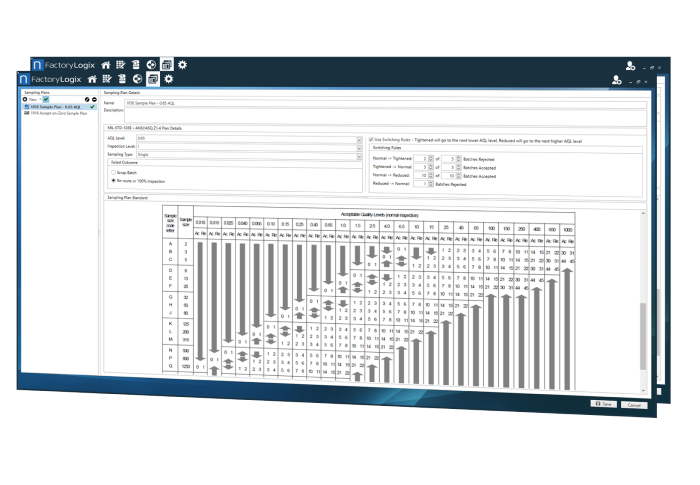
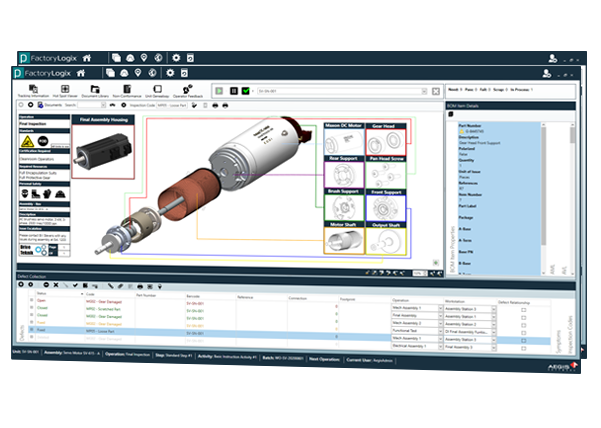
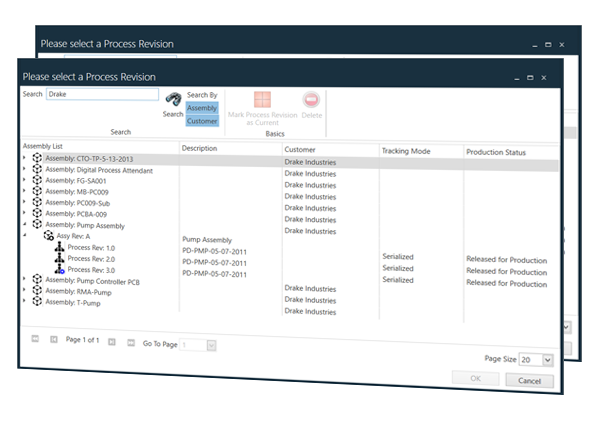
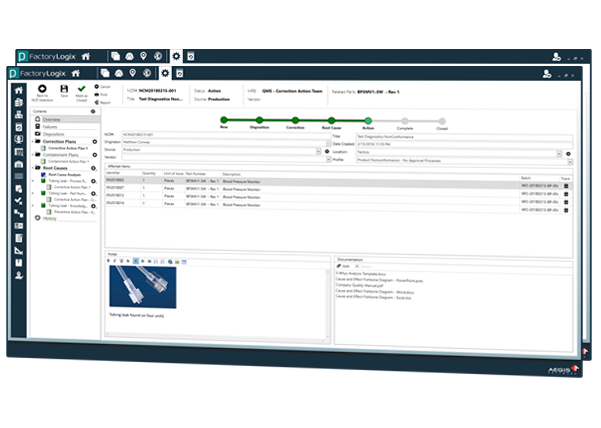
Automating Comprehensive Zero-Defect Quality
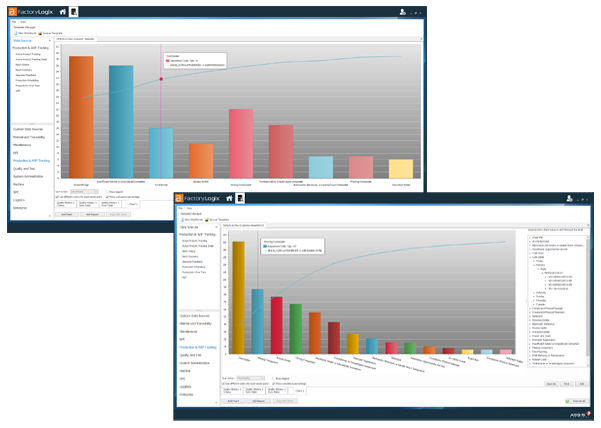
5% Reduction in Defective Parts Per Million (DPMO)
Improve First Pass Yield
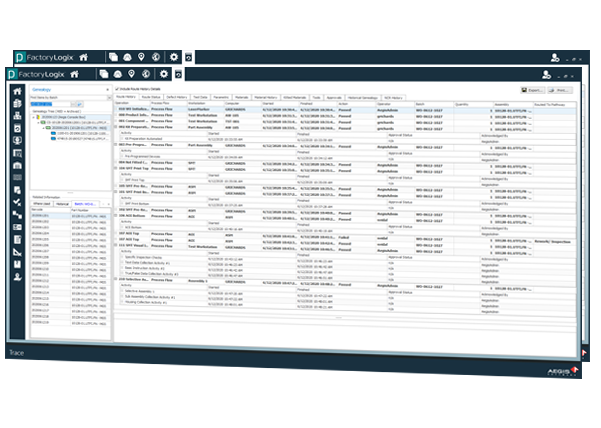
Achieve Level-4 Component Traceabiliy
Reduce Report Creation Time from Weeks to Real-Time
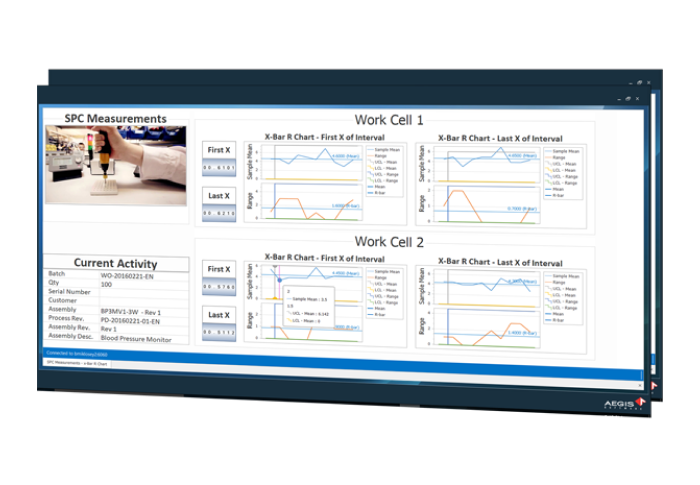
Reduce Assembly Rework Time by 30%
Improve Customer Response Times
SOME OTHER CLIENTS WE'VE HELPED